However, recent advances in single-cell RNA-seq (scRNAseq) provide a platform to track transcriptional changes across thousands of cells simultaneously. To understand development, and to provide a foundation for inventing therapeutics, we must comprehend the systems-level phenomenon of how genes and proteins are used across cell type and over time.Ĭharacterizing the in toto systems of a developing animal is a challenge. Furthermore, genes associated with genetic disorders are often poorly described in terms of cell and developmental stage. A major obstacle for regenerative therapies is not knowing the full system of genes normally expressed in a given cell type, thus preventing the development of cell-type specific genetic and pharmaceutical interventions as well as inhibiting the efficient reprogramming of stem cells to regenerate damaged tissue. Such coordinated gene expression programs are critical given that disruptions to the expression of cell-type specific RNAs can lead to abnormal function and the progression of degeneration and disease. A central question in biology is how gene expression unfolds over time to precisely coordinate the development of adult organisms. The specification of cell fates during development and the ability to function properly during adult life are determined by the RNA messages these cells express and the functions and quantities of proteins produced from these messages. The resulting atlas is a resource for biologists to generate hypotheses for functional analysis, which we hope integrates with existing efforts to define the diversity of cell-types during zebrafish organogenesis, and to examine the transcriptional profiles that produce each cell type over developmental time.ĭuring animal development, stem cells generate a vast diversity of differentiated cells to form functioning tissues and organs. Furthermore, we highlight the power of this analysis to assign new cell-type or developmental stage-specific expression information to many genes, including those that are currently known only by sequence and/or that lack expression information altogether. We annotated 220 identified clusters and highlighted several strategies for interrogating changes in gene expression associated with the development of zebrafish embryos at single-cell resolution. Our single-cell transcriptome atlas encompasses transcriptional profiles from 44,102 cells across four days of development using duplicate experiments that confirmed high reproducibility. To address this knowledge gap, we produced an atlas using single-cell RNA-sequencing methods to investigate gene expression from the pharyngula to early larval stages in developing zebrafish. Despite the crucial nature of this knowledge, we have yet to fully characterize all distinct cell types and the gene expression differences that generate cell types during development. The ability to define cell types and how they change during organogenesis is central to our understanding of animal development and human disease. (F-H) Replicate experiments for each developmental stage presented in this study are plotted together to show their relative contribution in the UMAP projection. Several representative clusters that do not show high correlation between replicates are labeled with their cluster number. We analyzed the correspondence between our replicates by counting the proportion of cells in each independent replicate, in every cluster and plotting these values in a scatterplot (x,y = proportion of all cells in a single cluster from replicate one, proportion of all cells in a single cluster from replicate two) and found a high level of correlation with R 2 values of 0.8 (1 dpf), 0.4 (2 dpf) and 0.8 (5 dpf). 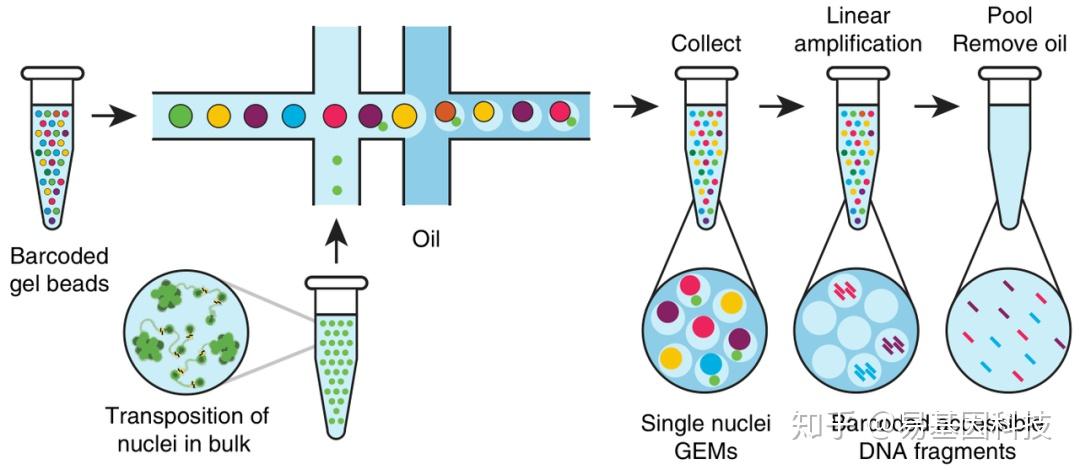
Replicate experiments for 1 dpf (C), 2 dpf (D) and 5 dpf (E) show mutual representation across clusters and indicate high reproducibility. (B) Summary of read, transcript, gene and cellular diversity and abundance for aggregated data used to produce the atlas. (A) The sample name, genotype, age, reads per cell and sequencing platform used for individual experiments. Summary of statistics related to 10x Chromium cDNA library preparation and sequencing.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
